|
FAQs about Giant Clam Disease, Pests & Predators
8
Related Articles: Example Chapter
from NMA Reef Invertebrates book, on Giant Clams, Tridacnids, A Brief
Guide to the Selection and Placement of Tridacnid
Clams by Barry Neigut,
Bivalves,
Mollusks, Lighting Marine
Invertebrates,
Related FAQs: Tridacnid Health: Pinched
Mantle Syndrome in Giant Clams by Dr. David Basti, Deborah
Bouchard & Barry Neigut, Tridacnid Disease 1, Tridacnid Disease 2, Tridacnid Disease 3, Tridacnid Health 4, Tridacnid Health 5, Tridacnid Disease 6, Tridacnid Disease 7, & Pest Snails (Pyramidellids...),
FAQs on Giant Clam Disease by Category:
Diagnosis,
Environmental, Nutritional,
Social, Trauma,
Pathogenic,
Treatments
Tridacnid Identification, Tridacnid Selection, Tridacnid Compatibility, Tridacnid Systems, Tridacnid Lighting, Tridacnid Placement, Tridacnid Feeding, Tridacnid Reproduction, Tridacnids 1, Tridacnids 2, Tridacnids 3, Tridacnids 4, Tridacnid Clam Business, Bivalves, Bivalves 2, Lighting Marine
Invertebrates,
|
|
Quarantine for new clams
8/17/19
Hey guys,
<Hey Jeremiah>
Thanks a ton for this site, it has helped me massively over the months I've been
involved with this hobby.
<Thanks for your words, we are glad to be helpful.>
I currently have a Maxima clam and am getting two more tomorrow. I successfully
kept the current Maxima for about two years and it has doubled in size and done
very well.
<Neat!>
I am planning on quarantining the two I get tomorrow in a ten gallon tank with a
sponge filter and heater. I plan on the duration of the QT to be 4 weeks as the
two I am getting are quite expensive and the one which I currently possess is
the prize of my tank. I plan on situating each of the clams I am getting in
their own piece of live rock (currently dry) on the bare bottom of the tank. The
pieces of rock I have picked out should be pretty good long term homes for these
guys. I will use a spare led light which is currently in my refugium to provide
them light. It worked great up top until the blue lights burned out and then
didn't look as good, but the white and other colors are what I need for the
photosynthetics anyway. One of the new clams is about six or seven inches and
the other is about two. I plan on filling the ten gallon tank, drip wise, with
water from my main tank as a drip acclimation method.
<Good technique>
So here is where I'm not sure about the next step. How much ammonia do clams
produce and how often should I change the water?
<They produce enough to quickly pollute small water volumes, especially when
recently imported.>
I know they consume Nitrates and that leads me to think that maybe they
shouldn't have water changes in the QT the same way a fish would. Is this true?
<Not completely true, Tridacna clams do consume nitrates but not harmful ammonia
and nitrites. And like any other living marine organism, they produce them...not
as much as fish, but they do, so you still have to do water changes during
quarantine.>
Assuming I do change the water, my plan was to drain off the water to an inch
above the largest clam and then drip water from my main tank into the QT to make
up the new water. I was planning on doing this once a week, but obviously if I
don't have to change the water I would rather not. I haven't found an answer to
this question in your archives anywhere.
<I suggest using a HOB filter filled with old filter material from your main
tank such as a filter floss or small pieces of live rock, anything that has been
for at least a month in your tank/sump, this way you will allow nitrification to
take place in your quarantine tank and ease the clams’ acclimation process,
besides you can get by with only once a week water changes, don’t forget to
constantly monitor your water levels.>
Thanks a ton for your time!
<Your welcome. Wil.>
Re: Quarantine for new clams 8/17/19
Thanks a ton!
<You’re most welcome. Wil.>
|
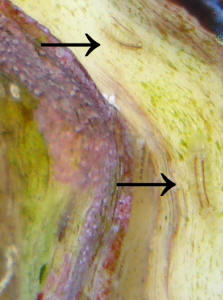
Can you ID these hitchhikers growing on my clam shell
please? 1/26/12
Hello Bob, James or
other possible responsive crew member,
<Jen>
I have attached some photos of some hitchhikers that must've
been on my clam when I got him. They started small and are
clearly growing. I have two other of these besides the three on
my clam in my tank attached to the live rock. They do not move.
Initially when they were little I thought they were Tube Worms or
a Vermetid Snail so I wasn't too concerned. Now I'm
wondering if they are Aiptasia Anemones
<They are>
or Hydroids. I'm not sure as they seem to
have characteristics of all of these. Please advise. It
doesn't appear that they are bothering anything. In fact, as
you can see in the photos no matter where I move the smaller
clam, he always finds his way right back to the bigger clam and
prefers to be adjacent to him for some reason. Anyway, It
doesn't look like these organisms were disturbing the mantle
on my bigger clam even though they were touching. I was concerned
because my husband had one of these hitchhikers on his maxima
clam in his tank, and when he removed it with tweezers
<Won't work>
(did not expose the clam to air) the clam later died. I think it
was related as the clam was doing fine, and there was nothing to
indicate any other reason it should've died. After he pulled
it out the clam stopped opening and died within a few days.
In the photos, the smaller clam on the left has three of them
growing out of his shell. The biggest organism is in the front
right on the clam in the photos and two smaller ones at the top
right on the other side of the clam.
I moved him to allow me to touch the organisms so that you could
see what they looked like when disturbed, and I wanted to try to
get a better photo.
I have a 70 gallon display with DSB and 40 gallon refugium with
DSB, Live Rock, rubble, Tisbe Copepods and Chaetomorpha Algae.
AquaC EV-180 protein skimmer, and I change carbon in a media bag
under the return for the protein skimmer every two weeks along
when I do water changes. I have a Wavemaker with Koralia Power
Heads, covered with mesh and sponge filters in the overflow box
and one power head as well on intake and outtake in the refugium
which I clean twice a week. Latest test results
Calcium 420
kH 10
phosphates 0
ammonia 0
nitrates 0
ph 8.4 lights on, 8.25-8.3 lights off.
temp 78 degrees
I recently started dripping Kalkwasser, and it seems to be
working out fine. It raised my PH slightly, but it was slowly and
now it's constant.
My calcium consumption rate is increasing as my corals and other
livestock are growing, so I test daily and dose if needed.
<I encourage you to read re Magnesium, test for this as
well... often
distorted ratios can cause troubles, in particular when
dosing/using Kalk>
My tank inhabitants include:
1 RBTA
1 Ocellaris Clownfish
1 Red Linckia Starfish (ordered a Fromia, but they sent me one
labeled as a Linckia)
1 Mandarin Dragonet-he eats most of the frozen foods I feed as
well as copepods
4 clams, One Maxima and three Crocea
Yellow Sun Corals
Orange Sun Corals
1 Scarlet Skunk Cleaner Shrimp
1 Blood Red Fire Shrimp
Hermit Crabs
2 Orange Spot Nassarius Snails
Bumble Bee Snails
Cerith Snails
Turbo Snails
A few LPS towards the bottom of the tank-one Scolymia, a colony
of Trumpet Coral, a purple Blastomussa, Dendrophyllia
Several SPS towards top of tank-Pink Birdsnest and Acropora
2 different Zoanthus sp. colonies
3 different colored Ricordea Florida Mushrooms
1 Hawaiian Feather Duster that has produced several more
Several yellow Feather Dusters that have multiplied as well.
I have two Orphek PR-156W LED light fixtures on a daylight,
moonlight cycle of 12 hours daylight, 8 hours moonlight, four
hours total darkness. While the display is in moonlight/dark, the
refugium T5's are on. I feed everyone a daily cocktail that
varies including frozen Mysis Shrimp, Brine Shrimp, Hikari Blood
Worms, Rotifers, Phytoplankton, Oyster Feast, etc. except the
RBTA which I feed 2-3 times per week with baby clams or raw
shrimp. I soak all my food with Selcon, and I recently ordered
Vita-Chem and will be including that in my foods as well. I
target feed the polyps on my Sun Corals daily. Even thought my
parameters are great and my water is clear thanks to the carbon,
skimmer, pods, filter feeders and Chaeto, I'm starting to get
quite a bit of algae growing on my live rock and equipment, so
I'm planning to add a small Zebrasoma tang to a quarantine
tank once it's cycled then to the display after 4 weeks to
take care of this.
I wrote a book as I want you to have all the information in case
I need to destroy these hitchhikers so you can tell me the best
plan of attack. I saw the preferred recommendations include
injecting vinegar, Joe's juice, buying a Nudibranch that only
eats anemones (but I have a RBTA) and Red Sea Aiptasia X.
<The last is my current fave>
I don't want to upset my well balanced setup.
<Mmm, perhaps better/best to just wait then... as you state,
the Glass Anemones don't appear to be mal-affecting
anything>
Thank You as always for your dedication, passion and information
sharing.
Jenny
<Welcome! Bob Fenner>
|
|

|
Re: re: Can you ID these hitchhikers
growing on my clam shell please? 1/26/12
Thanks Bob,
Do these anemones pose any threat being on my clam like that?
<Can; yes>
I've heard stories that they are ok at first, then become
aggressive as they get bigger. I read the reviews on Marine Depot
regarding the Red Sea Aiptasia X. Most are positive, but a few say
that the Aiptasia came back in droves. Have you found this to be
true in your experience?
<... see WWM re>
Also, will this product harm my clam or RBTA at all?
<Could; yes, but not likely if treated in situ>
Thanks for getting back to me so quickly and for all your help both
on your site and in questions. You are a very valuable resource and
have helped me an avid reefer. L
Jenny
<Ah good. BobF> |
|
Clam, hlth. 11/27/11
Hey Wet Web Media Crew!
<Gian!>
Wanna thank you all for your time, effort and patience, providing
everyone with all this information. Basically my question is,
there seems to be some sort of bubble growing underneath my clam
should i be worried about this?
<Mmm, yes... Involvements with the byssus can be
dire/deadly>
I believe the clam is a T. squamosa,
<I agree>
about 3 inches long, a friend had it in his tank for roughly 3
months, i was actually with him when he bought it, but apparently
has grown out of his tank (he has a nano) so i am the proud new
owner of this clam.
Parameters, although I'm not sure are needed for this
are:
Amm - 0
Ni - 0
Na - undetectable with api test kit
Phosphates - 0.00 with a Hanna checker
<Mmm, Tridacnids, all photosynthates, need some, measurable
Nitrate and Phosphate...>
(is this possible? i test my q/t tank with it as well and it
shows around 0.3 phos, so i guess it is working properly but
there is still algae growth in my sump so there must be
phosphates still right?)
<Yes and yes; though the algae may well be absorbing most all
soluble>
Ca - 480
Alk - 3meq/l
sa 1.025
mg - 1600 - should i try to lower this?
<Mmm, t'were it me, mine, I'd lower both Ca and Mg,
but if you're leaving, causing the Calcium to be this high,
I'd leave the Mg where it is>
ph - 8.1
temp - 77/78
Anyway, attached are a couple pics of the clam with the bubble it
is on the right side corner closest to the sps coral, my first
thought was some sort of vermetid snail, but now I'm thinking
it might be coming from the foot of the clam. Clam does not look
stressed in anyway, reacts normal when it is shaded, as in
retreats into the shell and i don't see any sort of gapping.
If a better pic is needed let me know, this is from my iPhone,
but i can get my hands on a digital SLR.
Thanks for your time,
Giancarlo
<Let's start w/ obvious questions. Is this Clam placed on
a piece of rock under the sand here? It appears to be too deep...
What is the PAR value where it's placed? What do you feed it,
the tank period?
DO read here:
http://www.wetwebmedia.com/ca/volume_4/V4I3/Tridacna/artGotTridacna.htm
and the linked files at bottom. Bob Fenner>
|
|
 
Re: Clam, Tridacnid, hlth. 11/28/11
So I read over the article, it has stated that T squamosa should
have rock underneath to attach the foot, as you were probably
going to suggest. Should I do this?
<Yes!>
I am assuming the clam has already attached its foot to the glass
at the bottom of the tank, would removing it do more harm than
good?
<Use a new single-edged razor blade...>
If not, what would be a good way to remove the clam so I can put
it on top of rocks?
Par value and lighting - I do not have equipment to determine Par
values,
<Look to borrow from a shop (LFS) or a hobby club, fellow
reefer>
but I think my lighting is more than sufficient, although it is
at the bottom of the tank, it is a very shallow aquarium, only 14
" in height, and after overflow and roughly an inch of sand
we are looking at closer to 11/12 " of water. I run a 250
watt Metal Halide, the bulb itself is a double ended phoenix
14,000K,
<Too high a temp. See the piece/article by myself and Calfo,
linked where you were previously referred>
along with one of those new LED Kessil 150A 15,000 K (which I
have been more than impressed with). Both hanging roughly a foot
away from the water. Bulb on the halide would be 5 months old,
and I change the bulb every 6-7 months.
Feeding the tank - I do not generally feed the tank much,
basically once a week for the past month I have been feeding reef
roids 1 teaspoon in about 120 gallons of water, I haven't
noticed much difference and I might just stop it all together. I
do however feed live Tisbe and Tigger pods, once a week each, I
have a mandarin dragonet so I will not stop. However I will start
to cultivate my own pods to cut costs, and after reading how easy
it is on your site it would be stupid not to. Finally for the
fish it is once a day either New Life Spectrum Flakes or sinking
pellets, and once a week frozen Mysis.
Right now I am assuming the best course of action would be moving
the clam, and placing it on top of some rock/rubble so it can
attach the foot.
<Nah>
If this would cause to <too> much stress, please advise as
I would suspect the foot has already attached to the glass and I
can only see that the removal of the clam would cause the foot to
tear.
Again, thanks for your time and help
- G
<Keep reading. B>
|
Stylochus
matatasi Flatworm... Clam et al. eater 11/16/11
Hello WWM,
<Meg>
By perusing your site, I have determined that we had & may still
have one or more of the Tridacna-eating flatworms in
our tank.
<No fun>
I lost 2 clams in July, but also many snails. Then in August, I caught
a HUGE flatworm. Grey-Brown, mottled. I figured it ate the snails but
did not ID the thing at the time or else I would have realized it ate
the clams too. We caught this thing and took it out but it broke into
pieces. I tried to pick up all the pieces (at night, in my pajamas,
holding a flashlight) but one may have escaped.
<Best to siphon out>
Forward to October. I still have 3 clams left. Start seeing dead snails
again. Then another clam dies. Two weeks later, another clam, and two
weeks after that, my last clam (this past weekend). All 5 clam deaths
in hindsight match exactly the MO of what happened to other
people's clams known to be eaten by a flatworm (by my
research).
<Did you treat this system w/ an Anthelminthic... an anti-worm agent
to rid it of this Polyclad?>
Took the last clam out and guess what crawled from his shell? A
flatworm. This time I ID'd it, it is the clam eater.
I used flatworm medicine and did WC...but I think the medicine is
really for planarians.
<Should work on/for all Platyhelminths...>
Will it kill the worm(s) I have?
<What is the active ingredient? Read here: http://wetwebmedia.com/fltwmchemcont.htm
and here: http://wetwebmedia.com/fshwrmidcuref.htm>
And how many do you think I could possibly have? Will I ever be able to
keep clams again?
<Should be>
Thank you in advance for your response!
Megan
<And you for sharing. Bob Fenner>Possible dying clam
8/26/11
I noticed this morning that my clam has not opened.
<<What type of Clam? Am going to assume for the purposes of this
response that it's a Tridacnid'¦but in the future please
be specific with the species if possible.>>
I can see just a small portion of the mantels inside the shell.
<<Is the mantle still on the brim or outer edges of the shell or
has it begun to wane or recede to the point where you can clearly see
the smooth lining of the inner shell?>>
I've had this clam for about 3 years (not sure of its age when
purchased - and don't really know their longevity in
captivity).
<<Again it depends on species, some fare much better in captivity
than others but three years seems relatively short. Many aquarists and
public institutions have Tridacnids that are decades old. Current data
suggests they do not perish simply do to age (giving them a great
potential life span) if they do perish it's usually due to
something environmental, disease or predation'¦the latter of
which most captive Tridacnids don't have to deal with if purchased
responsibly. There are reports of large T. Gigas surpassing the century
mark.>>
Should I give it a few days or remove it ASAP - I don't want to
pollute the
tank. It's a 125 display with 30g refugium. Water param.s are fine
(ALK=11;
CA=440; Phos=0; pH=8.3).
<<The parameters look good, though keep an eye on your alkalinity
as your 440 calcium ppm has a direct correlation with this, the more
dissolved calcium carbonate in the system the more difficult it will be
to maintain a stable balance of alkalinity, though if you're using
a reactor it likely is handling this ratio for you. As for the clam,
without more detail (or even better yet a picture) it is difficult to
diagnose the problem. >>
Would appreciate your advice.
<<You are correct in your worry that a deceased Tridacnid can
fowl up a system and it is best to watch for early warning signs and
remove it before it begins to decay in your system. I had to make a
similar tough call recently at a public aquarium I volunteer at with
one of our mid-sized T. Gigas, and eventually came to the unfortunate
conclusion that the animals chance of recovery was so low it wasn't
worth the risk of leaving it in the system to die and create organic
carbon issues. For checking out your clam I recommend a few of our
articles (but still would be more than happy to help you in the
decision making if you provided more details or a picture like I
mentioned above'¦and some more details about the tank as
well; other inhabitants, lighting, placement of clam, species of clam.)
If you start to witness animals in your tank that don't
readily/normally display a diet for healthy Tridacnids start to snack
on this clam (such as smaller fish and certain ornamental shrimp) this
means that they sense it is on it's last legs and are not attacking
so much as they are trying to recover the remains, if you notice this,
it's a good indicator that it's time to remove the clam if the
system isn't large enough to handle the decay.>>
<<More reading
http://www.wetwebmedia.com/ca/volume_4/V4I3/Tridacna/artGotTridacna.htm
http://www.wetwebmedia.com/BookMatters/WWM/NMA-RI/NMARITridacnidsdemo.pdf
http://www.wetwebmedia.com/ca/volume_6/volume_6_4/clams.html
http://www.wetwebmedia.com/ca/volume_6/volume_6_4/clams.html
http://www.advancedaquarist.com/2009/3/aafeature1
And this last one if you need assistance in id'ing your clam
http://reefkeeping.com/issues/2002-07/jf/feature/index.php
Regards,
<<To you as well.>>
-gene
<<Adam J.>>
Re Possible dying clam 8/29/11
Hello Adam,
<<Welcome back Gene.>>
Thank you for your response. My apology for not identifying the
clam.
<<No worries, it's just important to know the specifics so
we/I can further advise you.>>
It was a Crocea. Notice, I said 'was'. I decided to check the
stability of the clam knowing that if the foot was still well grounded
on the rock, it was likely still alive. As it turned out, it was not. I
removed it today.
<<Probably the best decision for the overall health of the
system.>>
What seems odd to me is the swiftness with which it died. I saw no
signs of distress until this morning. I do run a CR and rechecked my
param.s after writing you. CA is actually very high at 560 and KH is at
12. I recalibrated the pH probe and found pH at 8.07 not 8.3.
<<These environmental parameters would be my main guess, Calcium
carbonate levels are too high and affecting alkalinity and clearly your
pH. T. crocea is more sensitive than other Tridacnid species and when
things go bad for clams, they go bad quick..>>
Other corals seem fine and the fish show no signs of distress at this
time.
<<They are more resistant but given time will show issues as
well.>>
Of course, there could be other param.s that I can't measure that may
be in play here.
Again, my thanks for your informative reply.
<<Anytime...something else for you to read as well http://www.wetwebmedia.com/calcalkmar.htm
http://www.wetwebmedia.com/marphalk.htm >>
-gene
<<AJ.>>
|
Crocea Clam Issues/Tridacna Systems/Health
3/31/2011
Hey folks,
<Carl>
I have a question about my crocea clam who has been in my tank
for over a year now. As you can see in the picture, there are
spots (circled in red) on the mantle which appear bleached. It
has not always had these spots, though I can't really
pinpoint when they developed. In addition there on one side of
the shell there is an organism (circled in yellow) which is a
sort of transparent black color. You can see there are a number
of siphons coming off this thing. Is this an actinian?
<No, appears to be a tunicate colony. An actinian is a sea
anemone in the order of Actinaria.>
Its been there for at least 6 months. I tried cleaning it off
with a toothbrush when I first noticed it, but I probably
didn't get it all because it came right back. Neither issue
seems to bother the clam very much in an obvious way. You can see
from the second pic, there continues to be steady and constant
growth. The photo was taken first thing in the morning after
actinics came on, but before metal halide.
This particular reef tank is 65g with a 29g sump/fuge. It is
about two years old. Houses a mix of sps and lps corals and very
few fish (2 ocellaris, a Pseudochromis porphyreus and a yellow
watchman goby) . There's a clean up crew and lately we've
had a population explosion of snails and feather dusters. It sits
about 8 inches under a single 250w hqi MH at 10K, supplemented by
4 39w T5s. Right now I've got Wave Point Coral Wave bulbs in
there (very pink). <??> The clam itself is just over a foot
beneath the light source. This is a pretty low nutrient tank so
basic chemistry is consistently at 0 including nitrates and
phosphate. Daytime pH is 8.5, night time 8.1, dKH is 8. I have
not tested calcium recently because the only thing that gets
dosed regularly is Kalk from top off water that runs through a
Kalk stirrer. The tank is fed pretty heavily once a day and
sometimes twice with a variety of foods including rod's food,
oyster feast, some pellet and Nori.
The tank is basically thriving and the clam seems to be doing so
too. I just want to make sure these two issues are not of long
term concern but if so, what is your opinion on the action to
take. Also, will those bleached spots ever fill back in?
<I believe you problem lies in the sterility of your system.
Tridacna actually prefer and use nitrates in the water. Suggest
you read here and articles/FAQs found at the foot of the
article.
http://www.wetwebmedia.com/ca/volume_4/V4I3/Tridacna/artGotTridacna.htm.
And here.
http://www.reefkeeping.com/issues/2007-10/jf/index.php>
Thanks in advance!
<You're welcome. James (Salty Dog)>
Carl
|
|
 
|
|
Maxima clam doing very bed/ dying 1/12/11
Hi crew
My problem is the clam.
My system is 60 g hex, 154 w of t5 HO light fixture is 3"
from the water (2 marine Glo and 4 power Glo), 80 lb LR, 20 lb
LS, Fluval 4, remora hang-on AquaC, uv sterilizer 9w, chiller,
Aqualizer,
<The magnets?>
2-koralia2 powerhead, 1- koralia1 Power head , 1- polario 15 ml
wave maker,
Water condition: salt 1024,
<A smite low>
temp 77, nitrate 10, ammonia 0, phosphate 0,
<Umm, here's at least one fatal error. HPO4 is an
essential nutrient for Tridacnids...>
nitrite 0, calcium 470,
<High... what is your Mg?>
iodine 0.03, ph 8.2, kH 100
Stocked with 1- blue hippo, 1 -yellow tang, 3- yellowtail damsel,
1- four and 1- three-stripe damsels, 1- maroon clownfish, 1-
Dottyback bicolor plus some crabs and snails
Corals: green Sinularia, Candycane, torch , Galaxea, sun polyp,
maze coral, brain coral Favites, mushrooms, Zoanthids, red
Blastomussa, Echinophyllia, spider sponge and blue Tridacna
maxima clam.
I bought the 6 inch clam 3 months ago placed it on the rock work
about 7" from the surface of the water. The clam always
showed its mantel with nice colors but never opened fully. About
two weeks ago a flame angel started picking at it and the clam
retracted the its mantel and stayed closed, slowly trying to open
but the angel would bite it and he would hide again.
After about 6 days of abuse (week ago) I was able to remove the
angel and take it back to the Store. The clam looked better right
away until two days ago it shrunk into its shell and its not
moving. The shell is slightly open about 3/8 gap, I can see
inside it and observe a nice color but it's not
responding.
I have looked on the WWM and internet for information on this
dilemma with out a any result. I feel my clam has little time
left if it's not already gone. Please respond as soon as you
can. I have attached a picture of the clam as it looks right now.
Thank You
Pawel
<Mmm, I'd remove whatever chemical filtrant/s you're
employing... perhaps feed this animal directly. Bob
Fenner>
|
 |
Re: Maxima
clam doing very bed/ dying 1/13/11
Dear Mr. Fenner
Thank You so much for such a prompt response.
<Welcome Pawel>
Mg is 1200. Looking at my log HPO4 has always been low, too low (0
- .01),
<Ahh! Needs to be addressed. The chemoautotrophic algae in the
Clam require this absolutely>
calcium always too high (450 - 500) only recently (1 month) I was
able to get it lower and stable at 450.
<Again... this must be balanced/ratio with Magnesium
concentration, and alkalinity:
http://wetwebmedia.com/marine/maintenance/maintindex.htm
scroll down to the pH, Alk... tray and read>
The clam still looks the same and not moving, it's been three
days like that. I tried feeding it last night with no response .
What should I look for as signs of death?
<...? Please read: http://wetwebmedia.com/TridacDisF6.htm
and the linked FAQs files in the series above>
Thank You So much for Your help.
Pawel
<Welcome. BobF> |
|
Derasa Clam gaping? 3/22/10
To Bob and all who have helped me so much before,
First things first:
Nitrates: 0
<Need some>
Nitrites: 0
Ammonia: 0
pH: 8.2
Gravity: 1.024
Salinity: 33
Calcium: 400+
<Magnesium concentration? Needs to be proportional...>
So, after researching (on WWM, of course), and reading "Got
Tridacna", I learned that I could house a Derasa Clam under
my 260 watts of CF lighting in my 55 gal reef tank. My local LFS
had two of them for $89 each (if you
were to be a member of their "fish club") and I
purchased one. I drip acclimated him for about an hour and a
half, my LFS's pH and salinity were very close to mine. After
I had placed him about halfway up in the tank, 12" from the
light, I was observing him and noticed he was "gaping"
a little bit and I'm not sure if he was doing this at the LFS
or not. I went straight back to WWM and found that his mantle
shouldn't appear this way either. I trusted my LFS with the
thought in mind that I could return him within 48 hours.
<I would return this specimen>
So, what I'm asking is based on the attached pictures, is
this guy going to make it? Or, should I return him and make a
"healthier" investment?
<I would, yes>
Thank you so much for all that you do for not the hobbyists, but
the animals we are trying to keep as pets!
Paul in snowy Chicago
<BobF in sunny S. Cal.>
|
  |
|
Re: Derasa Clam gaping? --
03/23/10
Hey Bob,
Thanks for the timely response!
<Welcome Paul>
Since the first day that I got the clam, he's stopped the
"gaping" and actually seems pretty healthy. He opens
for the daytime and closes up at night, and when my fish cast a
shadow over him, he responds. Was there anything in the photos
that may have led you to believe that he might not be
healthy?
<Mmm, no>
Or was it just the issue of gaping?
<Yes>
Thanks again,
Paul
<BobF>
|
Please Help/Tridacna Clam
Health/Predators 3/18/10
You could not get rid of me for long,,, maybe a new thread.
<Hello Craig, and yes this will be a new thread.>
This morning I noticed two pyramid snails on my squamosa. I have a sad
face right now. After removing the clam I found about 10 more
underneath. Now I have a really sad face. Scrubbed all of them off and
returned. After inspection of the disgaured <discarded> water I
could also see three of what I could only describe as a really small
round flat clear worm, about the size of a pencil lead?????
<No idea here without a pic. Bob and/or crew may input here.>
Any way back to the devil snails, I am in trouble aren't I?
<Not yet.>
I also have a big derasa about 5" or so and could not find any on
him, he is attached to a old clam shell so I dont know if this helps
these snails from getting underneath. Both clams are on substrate.
<Are you sure these are Pyramid Snails? Pyramid Snails are generally
white in color, have a slender shell that tapers to a point, and do not
grow larger than a few millimeters. The Pyramid Snails use their long
proboscis, which is a tube-like mouth, to puncture the mantle of a
Tridacna Clam in order to feed upon the fluids and zooxanthellae cells.
A few of these snails will typically not pose a threat to a healthy
Tridacna Clam. If not controlled/eliminated, the snails can reproduce
every 90-120 days, and if left to multiply, can cause serious harm or
even death to the clam in a very short period of time.
If the infected clam has poor expansion and loss of color within the
mantle, it may be too late to help it. Pyramid Snails feed mostly
during the nighttime hours, and during the daytime, they will remain
either at the base of the clam, or on the folds of the shell where they
are difficult to spot. It is best to inspect with a flashlight a few
hours after the lights have turned off on your aquarium. The snails
will line up at the top of the shell just below the clam's
mantle.
As you mentioned, it is best to remove the clam to a separate container
of aquarium water where you can physically remove the adult snails, and
brush the entire shell with a new toothbrush in order to remove any
eggs.
I would do this weekly until no sign of the snails exist.
I will add that natural predators of Pyramid snails include several
species of wrasse within the genus of Halichoeres and Pseudocheilinus.
A few of the more popular species that are reportedly very efficient at
controlling these snails are the Six and Eight Lined Wrasse (P.
hexataenia and P. octotaenia), and the Green Wrasse (H. chloropterus).
Hoping this helps remove your sad face. James (Salty
Dog)>
Re Please Help/Tridacna Clam
Health/Predators 3/18/10-3/19/10
Thanks again, after getting home about 7 est the Squamosa, believe it
or not is the fullest and most beautiful I think I have ever seen. I do
not know if this is because he may had been bothered for some time by
these pests? and now there gone he is happy again? I do believe they
are pyramid snails by pics and also where I first spotted the 2-3 up by
it's mantle.
<OK.>
I forgot to tell you that I lost a maxima 2-3" about a week ago,
but for some reason he never really seemed happy in my tank, so I did
not check to see if it was a snail problem, and forgot to mention it.
Have these guys always been in my tank?
<Likely came in with one of the clams.>
I have had the maxima, now dead about 3-4 months, the derasa about the
same and squamosa around 2.Also when I cleaned squamy I notice a slimy
layer of ??? which after doing some research sounded like what their
egg masses are like. Do they only lay eggs on host?
<Yes.>
I have never seen them on the clam after 1-2 hours of lights being off
only this morning about 3 hours before lights are to come on, and two
days ago my led moon light went out so now tank is in complete
darkness, could this excel the problem? Meaning they did not come out
of hiding because of the moon lights being on at night.
<Unlikely, there are full moons over the reefs.>
As far as the six line, I have a mc coskers <McCosker's> now
are they compatible?
<Compatible with whom, or do you mean will the McCosker's Wrasse
eat the snails? If so, I haven't heard any reports of this wrasse
eating pyramid snails.>
and I read they may not get rid of them because of their sleeping
habits at night time when the snails attack.
<The snails can/will also burrow into the sand bed near the clam and
is where the wrasses will find them.>
Are clams their only food source?
<Likely so, but not 100% positive. Bob or another crew member may
input here.>
Meaning in worst case if all clams die, I would just have to wait out
their reproduction cycle to introduce clams again?
<If your clams are healthy now, there is no reason they should die
from pyramid snails as long as you remove the snails and scrub off egg
sacs as you have been doing. Eventually, you
should eradicate them completely and I would suggest getting a Six Line
Wrasse to help you out.>
As far as the worms, To <too> small to take a pic, after a close
look, looked like a round clear stingray no tail, instead of two eyes
looked like a brown dot. One more why don't they go after my nice
juicy derasa? Is the shell he is attached to help in any way?
<Possibly, depending on the clams location. Most folks that have
Pyramid Snail outbreaks will temporarily place their clams on live rock
once they are cleaned of the snails and eggs to prevent re-infection.
Pyramid Snails do not generally leave the sand bed for higher ground
other than feeding on a Tridacna.>
thank you, I will talk to you soon, have a great eveing
<evening>. I am off to kill these little ba%%((e((reds.
<That's the spirit, and can I please ask you to run future
queries through a spell checker? Will save me much time if I do not
have to edit/correct before posting on the dailies.
Thank you. James (Salty Dog)>
Re Please Help/Tridacna Clam
Health/Predators 3/18/10-3/19/10-3/22/10
I am sorry, since my accident, I need help with a lot of things.
<Sorry to hear this, Craig. James (Salty Dog)>
Craig
|
Derasa clam, hlth., paucity of facts
3/1/10
Hello WWM, love the site. I can spend hours reading all the
replies. One question I am concerned about my clam, it has some
curled in mantel, I ordered him from a wholesale supplier and it
looks to be wild caught.
Anyways to the point is this a sign of pinched clam?
<Is it this way all the time?>
Here is a attached picture. He/She looks like it has been nipped
by a few fish.
<Yes... what else is present here livestock wise?>
I have had him in for a week
<Oh, may be "just new" to your conditions,
system>
seems to open fine every mourning and close at night. I suppose
the all important tank stats are as follows nitrate=<10
nitrite and ammonia=0 ph=8.4 calcium is 460.
<A bit high... how do you adjust this? And very importantly,
what is your Magnesium concentration and Alkalinity? Please see
WWM re these important measures, co-factors>
This is an a 30 tall
<Not very large... inherent issues, instability...>
with 4 t5 ho and 175 MH I change 3 gallons of water every Sunday
with Seachem reefsalt. the only thing I supplement is marine snow
twice a week.
<Mmm, almost worthless. I would not use this product.
Non-nutritive. But... what else do you feed the clam/system? Has
this system been set-up long, have an established good-sized
refugium?>
Thanks for the help
<Need much more useful information to aid your efforts. Do
read through the articles, FAQs files archived on Tridacnids on
WWM:
http://wetwebmedia.com/HighInvertInd.htm
Bob Fenner>
|
 |
|
Crocea Suicide Attempt... Tridacnid placement f'
-- 02/08/10
Hello folks,
<<Greetings Lanny>>
I just awoke this morning and checked my 30g/150w MH/54w T-5 that
is in my bedroom. To my dismay, my Crocea had hurled itself off
the rockwork and smack on top of some pesky Actinodiscus.
<<Uh-oh>>
I am not so much worried about the sting as I am about the
strange bundle still attached to his rock.
<<Mmm, yes'¦damage to the byssus gland can prove
fatal>>
At first I thought it was the byssus gland, but as I read, it
seems more likely the byssus fibers.
<<Okay>>
She seems alright...the foot looks unscathed and the mantle is
expanding. I have included a pic of the bundle - my apologies for
the amateur photography. Am I correct in my assumption that is it
merely the fibers and not something worse?
<<From the pic it appears to be more than just
'threads''¦but if the 'clam initiated the
move,' as opposed to being 'knocked off' the rock,
then it is likely fine'¦though only time will tell.
And as it should be obvious, the clam was not happy in its
previous location. I think it unlikely this species was receiving
too much light here, so do look for other reasons for its wanting
to move (encroachment from corals/other stinging organism, too
much/too heavy water flow, etc.)>>
Thanks for the help and all your great resources.
<<A collective effort'¦you're quite
welcome>>
Regards,
Lanny
<<Cheers'¦ EricR>>
|
 |
|
Re: Crocea Suicide Attempt... --
02/09/10
Thanks for the quick response.
<<Quite welcome>>
The clam is in fact looking great today. The mantle is fully
extended, and the portion that was touching the Discosomas looks
unscathed.
<<Excellent>>
The clam was contacting a Montipora capricornis in its original
spot, but they had grown into one another maybe three months ago.
Could that be the irritant?
<<Possibly...>>
The mantle would rest on the Monti when it was extended. I have
never had trouble with Acroporids irritating my clams in the past
so I thought nothing of it. This clam actually used to reside
nestled into an Acropora colony with no evident nettling.
<<Though some have stated that these clams (Tridacnids) are
very 'tolerant' of contact with other cnidarians, I have
to think there is a 'limitation' to that
contact'¦perhaps variable among the differing species
and even among different specimens>>
I also wanted to inquire if the scutes that were broken in the
fall have the potential to harm the mantle.
<<Sure'¦if jagged/sharp enough>>
They are sort of jagged. I hate to sound like a mother with a
newborn, but I worry about all my critters.
<<There's likely little reason to worry, but if these
breaks are overly sharp/pointy, you could remove the clam from
the water (make sure it hasn't reattached to the substrate
first) and try to file them down a bit with a 'fine cut'
metal file'¦or better yet, one of those small fine
'diamond-grit' files used to sharpen carbide router bits
and the like>>
I see too much slaughter working in the aquarium industry here in
So Cal. Last week I saw a 6' black-tipped reef shark crammed
into a tank at All Seas and it cut deep.
<<Sad indeed>>
Thanks again for the help.
Lanny
<<Is my pleasure'¦ Eric Russell>>
R2: Crocea Suicide Attempt... --
02/16/10
Hello again,
<<Hi Lanny>>
Just thought I would give you the update on the Crocea.
<<Great!>>
It has been about a week now, and my clam is doing great.
<<Ah!>>
After the jump, I placed the damaged clam on a piece of Montipora
skeleton (another falling incident...Monti cap onto
Fungia.--Monti lost...bad) and put it on the sand, front and
center. I could actually see clear through from the incurring
siphon through the foot and out the other side, and I was a bit
worried. The mantle never stopped extending fully, and it even
has repaired some nicks in the scutes.
<<A good sign>>
While shifting things around to accommodate a couple new frags, I
gingerly moved the Crocea to find that it had already repaired
itself enough to attach to the base!
<<Very encouraging indeed!>>
Thanks again for the help.
Lanny
<<I'm pleased to hear the clam is doing so well my
friend'¦ Cheers, EricR>>
|
05/02/10 Clam and Hammer Coral comp.
Hello:
<Hi Megan>
I introduced a hammer coral into my reef tank last week. Coral doing
great, but my maxima seems bothered now.
<Mmmm>
He has gone from upright on his perch to leaning sideways as of this
AM...the side closest to hammer coral is what he is trying to move
farther away.
<I see>
It's not as if he is right next to coral...hammer is at bottom, he
is at top on rocks...at least 8-10 inches of space between them....but
he definitely seems irritated...could the hammer's chemicals
flowing thru the water be upsetting him?
<Yes, and this coral has been reported to have very long sweeper
tentacles>
Can I wait until tomorrow to take the coral back to LFS?
<Probably yes, but is the Clam 'downstream' of the
Euphyllia? If so, then switching these so that the sweepers are pushed
in the other direction might do the trick>
If I have to choose, I choose the clam!!
<Move one of these and observe>
Finally, what corals will not bother my clam? I have Zoanthid,
Mushroom, Star Polyps, etc. but wanted something a little more
advanced.
<Mmm, try something like a Montipora, an excellent, non-aggressive
intermediate coral, that is usually ok with what you have
listed>
I did read up on this stuff, I do ask lots of questions...but these
spur of the moment situations sometimes throw me for a loop...thanks
for your time!!
<No problem Megan>
Sincerely,
Megan
<Simon>
|
Amphipods Irritating Maxima clam? 12/7/09
Hello
<Kevin>
I am having some concerns over one of my maxima clams not opening
completely. At first I thought it could be pinched mantle. I have
researched this and don't find a whole lot out there on the
subject.
<Oh! Barry Neigut just had our online 'zine run his
definitive piece on this ailment:
http://wetwebmedia.com/ca/volume_6/volume_6_4/clams.html>
I am also hesitant to do the prescribed freshwater dip which
could possibly kill an already stressed clam. Especially since I
am uncertain this is what it could be. When the lights are out in
my tank I searched with a flashlight and noticed amphipods all
over the outside of its shell. Attached is a picture pointing a
few out.
<Well done!>
Placement is in the base rocks in sand. I believe they are
amphipods after searching this site and many other
references.
<Do appear to me as thus>
I am wondering if they are irritating the clam causing it not to
extend its mantle fully.
<Could well be>
I also notice a few crawling about during the day which leads me
to believe this could possibly be the case. I have
another maxima which looked very good for a while now showing the
same signs. Amphipods are all over this shell as well. I have a
crocea clam the amphipods don't seem to bother which looks
great. Maybe moving the maximas
higher up away from the sand would solve the problem?
<Mmm, doubtful... I would...>
Also nothing else in the tank seems to bother the clams (2 true
percula clowns, hippo tang, Kole
tang, Banggai cardinal, 2 cleaner shrimp)
My calcium is 380 (working to get this to at least 400).
Alkalinity is 11dKh. Magnesium is 1200 ppm. pH is 8.2. Any
insight would be great.
Thanks in advance.
Kevin
<I would seek either to bait/trap them out (meaty food wrapped
in some filter media, stuffed in a plastic pipe... at night)
removed after an hour or two), or look to "renting" a
relatively, most likely non-Tridacnid-predator that will hunt
down, eat these. The list is long here; I would sort through a
search on the Net in general. Bob Fenner>
|
 |
|
Re: Amphipods Irritating Maxima clam?
12/16/2009
Dear Mr. Fenner,
Wanted to thank you for your help with my Maxima. I removed the
clam from the sand to move higher in the rock work. I decided,
while it was detached, to give a freshwater dip knowing after I
placed it in the rocks it could be challenging to remove if
necessary. As soon as the clam was placed in the dip water the
amphipods on the clam fell off.
<But fast>
There were quite a few more than I had actually thought. After 20
minutes I returned it to the top 1/3 of the tank and am happy to
report the clam looks 100% better. It has attached, has been wide
open and has been for about a week. Thanks again for your
help.
Best,
Kevin
<Ahh! Congratulations on your success, and thank you for your
report here.
BobF>
|
Compatibility... Bivalves and Crabs... feast
11/29/09
Hi.
<Hello Sony>
I had a weird issue, I had a maxima clam some smaller green clams a
flame scallop and they all got eaten after in introduced some blue and
red dwarf and zebra crabs, along with some Nassarius and margarita
snails . could
any of these have eaten all my clams. who and why? and does this mean I
cant ever add clams because I wont be able to find the culprits and
round them all up?
<Very little info for me to base an informative reply on. I would
like to know your tank size, water parameters, lighting make-up, and
what types of fish and other invertebrates, if any, are in the
system.
James (Salty Dog)>
Sony M. Onatte
Re Compatibility, Take 2, now reef... env. issues
-- 12/01/09
I have a 30 gal long with roughly 45 - 50 lbs of live rock. water
levels all normal.. 0 nitrate nitrite and ammonia. salinity 19-22.
<1.019-1.022? This should be much closer to 1.026...>
If everything is 0 do I still have to do water changes cuz I'm
topping off water 3x a week cuz it evaporates so fast.
<Short answer: yes. Long answer -- the fresh water you are adding
back 3x a week is not equivalent to a water change -- this is only
replacing the evaporated system water. No export of nutrients has
occurred. Water changes achieve 2 goals: nutrient export from the tank,
and replenishment of ionic balance within the water. These two things
are indispensable, and cannot be substituted by any filter, additive,
or otherwise.>
My lighting system is a Current USA power compact with Dual Daylight
Lamp (6700K & 10,000K), 1 Dual Actinic Lamp (420nm/460nm) and 3
Lunar Lights.
<Not nearly enough intensity for clams -- Power Compacts simply
don't have the depth of punch that T5 or Metal Halide
offer.>
<I> have an <A>qua <C> <R>emora
skimmer<,> <I> have a black clown<,> a
reg<ular> clown<,> a domino, 1 chromis, 2 Fiji blue and
yellow devil. 3 camel shrimp, 2 skunk shrimp, assorted margarita,
turbo, and Nassarius snails. 3 emerald crabs, 25 assorted blue and red
dwarf crabs. 5 diff types of Zoa's.
<Sounds nice -- a bit overstocked... but please, in the future do
run your email through a spell check, practice proper punctuation and
capitalize proper nouns... All is posted for posterity here, and these
corrections
take time out of our day.>
2 bleached trumpet corals from 3 months ago because one of my pumps
died while I wasn't home for a month. ( will their color ever come
back or is there anything <I> can do about it)
<If they have not died yet, there is hope.>
an recently eaten maxima<,> assorted green clams<,> 2 flame
scallops. <Flame scallops do not fare well in home aquaria -- and
'assorted green clams' gives us nothing to go on here..>
and PS: my Zoa's aren't growing as fast as they once were
almost to a stop.
<Your salinity is far too low, your ionic balance is off -- fix
this, your growth will increase.>
any magic food? I rarely target feed. hopefully this is good enough
info.
thank you
<Should not need much/any direct feeding in an adequately equipped
configuration, but would certainly benefit from a feeding of
Cyclop-Eeze or other similar small foods.>
Sony M. Onatte
<JustinN>
Re: Compatibility... Now ScottV rallies!
12/1/09
I just feel like the problem hasn't been addressed.
<It has by Justin below, water quality, stability and lighting are
the culprits here, not the crabs.>
My maxima clam has been in my tank for over a year, the green
iridescent small clams were newer but they were fine too until I added
a clean up crew.
<Unfortunate coincidence.>
All the dwarf crabs I was talking about. Then the clams just started to
clear out. No flesh left in the shell. I wanted to know if you have
heard of this before?
<Yes, the crabs are scavengers, they will feed on dead or dying
clams.>
And will I ever be able to have clams because of this?
<Sure, by addressing the issues listed below! <<Above now!
RMF>> Scott V.>
Unhappy Clam?/Tridacna Health 9/9/09
Hey gang!
<Hello Carla>
Quick question, my t. crocea clam just detached itself from the rock
it's been happily (or so it seemed) anchored to for over two years
now. I had noticed the clam "rocking" itself on occasion,
usually when stressed such as when the lights first come on in the
afternoon (I'm on a reverse light cycle to help control temp.
swings- the tank gets no natural light).
Otherwise seemed ok, good mantle extension, etc. Obviously it's
unhappy about something though, or wouldn't have jumped ship like
that! Didn't notice any byssal threads in the tank, attached to the
clam, or the rock. I moved the clam back up to his perch on the
rockwork, and redirected the powerhead that had been blowing behind
that rockwork in case that was irritating him- his spot is also below
the intake of the skimmer, but there are no discernible strong currents
that I noticed when my hand was in the tank. Any other tips? Just keep
an eye on 'em? It is just beginning to extend its mantle a bit-
obviously clamped shut for a few minutes after moving it. I wonder if
it is outgrowing the shelf/nook in the rockwork and was trying to find
more room to grow? I have read that it's a good idea to leave the
clam on the sand if they detach with a piece of rock below them in the
sandbed to attach to, but this is a 29 gal. tank with PC lighting (60
watt 50/50, 60 watt 10000k?) so it needs a high placement in the
tank.
<For sure.>
The bulbs are in need of replacement (a little over a year old, I
believe)-
could the wavelength shift with aging bulbs be part of the problem?
<Possibly, as your lighting is close to borderline to start with.
Anyway, wondering if you had
any feedback and I'll keep you posted on any new developments!
<I would replace the lamps and further observe. A link to a very
informative clam article by James Fatherree, a regarded clam expert,
will be good reading/learning for you.
http://www.reefkeeping.com/issues/2007-10/jf/index.php
Thanks!
<You're welcome. James (Salty Dog)>
*Carla
Re Unhappy Clam?/Tridacna Health
9/18/09
James and Crew,
<Carla>
Thanks for your advice!
<You're welcome.>
I moved the clam back to his original position in the rockwork, only to
have him "jump ship" again by the next morning, so I perched
him in a smaller piece of live rock that is a thickly encrusted shell
of some kind, and has a nice hollow cup for him to sit in, where he
seems quite happy and has attached, and is now at least 2" closer
to the lights.
<Good, now leave it alone.>
When I turned this rock over to repurpose it for a clam perch, I
discovered a tiny light-starved mushroom polyp growing on the underside
(now the top)- this is growing next to the clam- will it irritate or
injure the clam? If so, suggestions to relocate the polyp? Careful
razor blade surgery? It's a quite smooth surface, so might work,
and I'd obviously rather sacrifice the polyp than risk damage to
the clam... but thought I'd leave the two for a bit to see if they
can work things out, unless anyone has advice to the contrary...
:-)
<Leave alone, not to worry until you need to.>
Several people have commented- "A piece of your clam fell
off!", which of course led to minor panic on my part until I
realized the were talking about the mushroom anemone, which has similar
coloration to the clam, and is attached to the rock near the base of
the clam. Funny...
<Yes.>
I also discovered the root cause of clam distress- stupid stupid stupid
me neglected to plug in the canister filter after cleaning it
(don't do vital maintenance tasks when distracted by company next
time, I suppose). Ack!
Opened it up, and man was that stinky- sent my fiancé© and
his friend practically running out of the room. Discarded the media
(except for the Chemi-pure, which I rinsed/soaked very very well-
should this be discarded as well?
<Not if it is still functional. Water clarity will determine that. A
good test to determine this is to put several stripes on a white paper
with a yellow highlighter, then tape the paper on the side glass.
While looking at it from the opposite side glass, you should be able to
see the yellow highlighting. If not, time to change the Chemi-Pure. I
prefer to change the Chemi-Pure before this stage, say every three
months with
an average stocked tank.>
Obviously has lost a lot of effectiveness, but it won't leach
contaminants back into the water, correct?),
<Shouldn't.>
added carbon and floss to temporarily polish the tank (usually I just
run Chemipure or sometimes PolyFilters as well, and mainly use the
canister for increased water volume and circulation), and filled with
new salt water, and within 24 hours everybody in the tank looked much
happier- I'd wondered why the skimmer was suddenly producing double
the usual amount of goop. Sigh... live and learn, I suppose... bad tank
owner!
<No, just the learning curve being experienced.>
Question about a new addition- bought a serpent star on Tuesday (and a
4 # hunk of gorgeous cured live rock with a pretty polyp on it... no
one believes me when I tell them the rock was more expensive by several
times than the starfish.. heh), and he's still hiding out in the
live rock cave he retreated into when I released him.
<Far more active at night.>
There was quite a difference in salinity from the store tanks (1.028 in
the bag) and mine, which is around 1.023... working to bring that up
slowly with water changes and topping up with light salt mix instead of
fresh water. I drip-acclimated him for just over 6 hours (even though
the LFS guy said that I didn't need to drip-acclimate- sounded
crazy to me and contrary to everything I'd read... did it
anyway)
<LFS is wrong here.>
to help even out the dramatic shift in salinity etc, but know that
these animals are quite sensitive.
<Sensitive to sudden pH/salinity changes, correct.>
It's definitely alive, and looks healthy, but sluggish.
Patience?
<Yes.>
He was the most active guy in the store tank, climbing the walls with
the lights on. I have my tank on a reverse-light cycle, and was hoping
to watch his "nocturnal" scavenging activities... hopefully
he just needs time to settle in and recover from the stress of
acclimating to new tank conditions? Anything I can do to help him
out?
<You are doing fine.>
Thanks for all the reassurance and advice that I've gotten from you
guys over the years- what a great resource!
<You're welcome. James (Salty Dog)>
Cheers,
*Carla
Clam with hitchhikers -- 09/08/09
Hi
I hope everyone is well at the WetWeb!
<I am for sure!>
I recently purchased a 2.5 inch Maxima clam, with 2 large, and 3
smaller clove polyps on its shell. One of the large ones is at the very
top, and the mantle of the clam expands around it to open, the others
are further
down the shell, (and obviously reproducing).
The Clam seems ok - it opens fully, but I don't know if I should be
removing these polyps or if that would be a worse stress on the clam
than leaving it alone. If so, how would you recommend (Exacto-knife was
my best guess...)
Thanks as always,
Karen
<A sharp blade, yes... not likely chemically toxic or physically
harmful however. Bob Fenner>
|
|

